Highlights
- Kyoto University researchers develop SEEE process to recover high-purity rare earth elements from used magnets.
- Lab results show 96% neodymium and 91% dysprosium recovery rates using molten salt electrolysis.
- Technology is promising but not yet commercially viable, requiring further industrial scale testing and economic analysis.
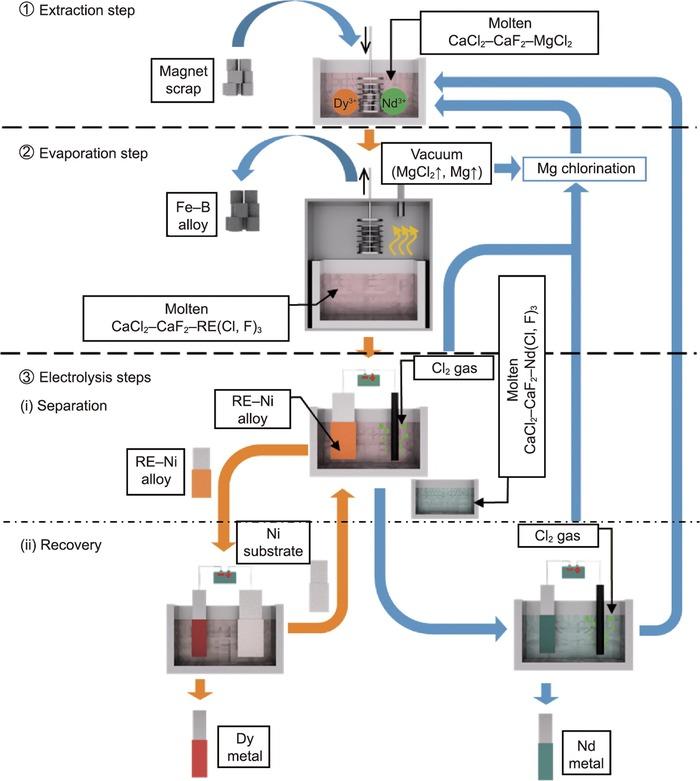
A recent Newswise article (opens in a new tab) touts a “transformational” breakthrough in rare earth element (REE) recycling from Kyoto University (opens in a new tab). The featured study unveils the SEEE process—Selective Extraction, Evaporation, and Electrolysis—designed to recover high-purity neodymium (Nd) and dysprosium (Dy) from used magnets. The research team, led by Professor Toshiyuki Nohira (opens in a new tab), reports recovery rates of 96% for Nd and 91% for Dy—figures that, if scalable, could meaningfully reduce waste and dependency on fresh mining.
So far, so good: the facts as presented appear accurate and are supported by a peer-reviewed publication in Engineering (DOI: 10.1016/j.eng.2022.12.013 (opens in a new tab)). The process uses a molten salt mix—CaCl₂ and MgCl₂—with calcium fluoride to limit evaporation losses. Electrolysis separates the elements based on formation potential. It’s a clever, elegant approach to a longstanding industrial problem.
What’s Vapor, What’s Vision
Here’s where things drift into optimism-as-narrative. While the lab results are impressive, the article glosses over the known chasm between bench-scale and commercial-scale viability. There’s no cost analysis, energy use estimate, or lifecycle comparison to existing recycling or hydrometallurgical methods. These are key metrics any institutional investor—or industrial user—will demand before taking the process seriously.
Additionally, the article’s claim that this technology will “transform green technology” and “boost carbon neutrality” borders on the messianic. Transformational? Maybe. Proven as a viable industrial route? Not yet. This leans science journalism by press release: high on promise, light on risk or commercial barriers.
Limitations
Rare Earth Exchanges (REEx) reviewed the study and identified the following limitations:
- The process is not yet scaled or piloted for industrial deployment.
- Electrolysis current efficiencies and Dy/Nd separation ratios are concentration- and potential-dependent.
- Recovery of Dy at high purity depends on achieving a Dy/Nd alloy ratio >9, which is only possible under specific lab-controlled conditions.
- Evaporation losses of REEs remain nontrivial, even with CaF₂ added to suppress vaporization.
- Industrial readiness is not demonstrated, and no techno-economic or lifecycle analysis is included.
Investor Lens: Encouraging, but Don’t Bet the Smelter
For rare earth investors, the SEEE process is not yet a market-mover, but it’s a signpost. Efficient REE recycling is a critical part of closing the loop in magnet and EV supply chains. Kyoto University has produced one of the more scientifically rigorous solutions to date. But until pilot plants emerge—and until energy and cost equations balance—this remains pre-commercial innovation, not a disruption.
As always, Rare Earth Exchanges (REEx) recommends grounding excitement in engineering fundamentals, not clickbait headlines. SEEE may indeed shine—but it’s still early days in the furnace. But we’ll keep monitoring this development.

0 Comments