Highlights
- Researchers achieved 93% recovery of neodymium and praseodymium from discarded hard drive magnets using hydrochloric acid processing, offering a viable urban mining alternative to traditional rare earth mining.
- Three recycling methods were compared: HCl leaching (93% recovery), sulfuric acid (73%), and biodegradable chitosan-EDTA biosorption, revealing an efficiency vs. sustainability trade-off.
- While lab results are promising for reducing Western dependence on Chinese rare earth supply, scaling to industrial production faces cost, environmental, and regulatory challenges.
A new study led by Muhammad Daim Abbas (opens in a new tab) of the Institute of Space Technology (Pakistan (opens in a new tab)), in collaboration with researchers from King Saud University (opens in a new tab) and the Chinese Academy of Sciences (opens in a new tab), demonstrates that rare earth elements—specifically neodymium (Nd) and praseodymium (Pr)—can be recovered from discarded hard disk drive magnets at rates as high as 93% using hydrochloric acid (HCl) processing, with alternative biodegradable methods also showing promise. For lay readers: this means valuable materials critical to electric vehicles, wind turbines, and electronics can be extracted from old devices rather than mined anew—offering a potential secondary supply stream in a world heavily dependent on China.
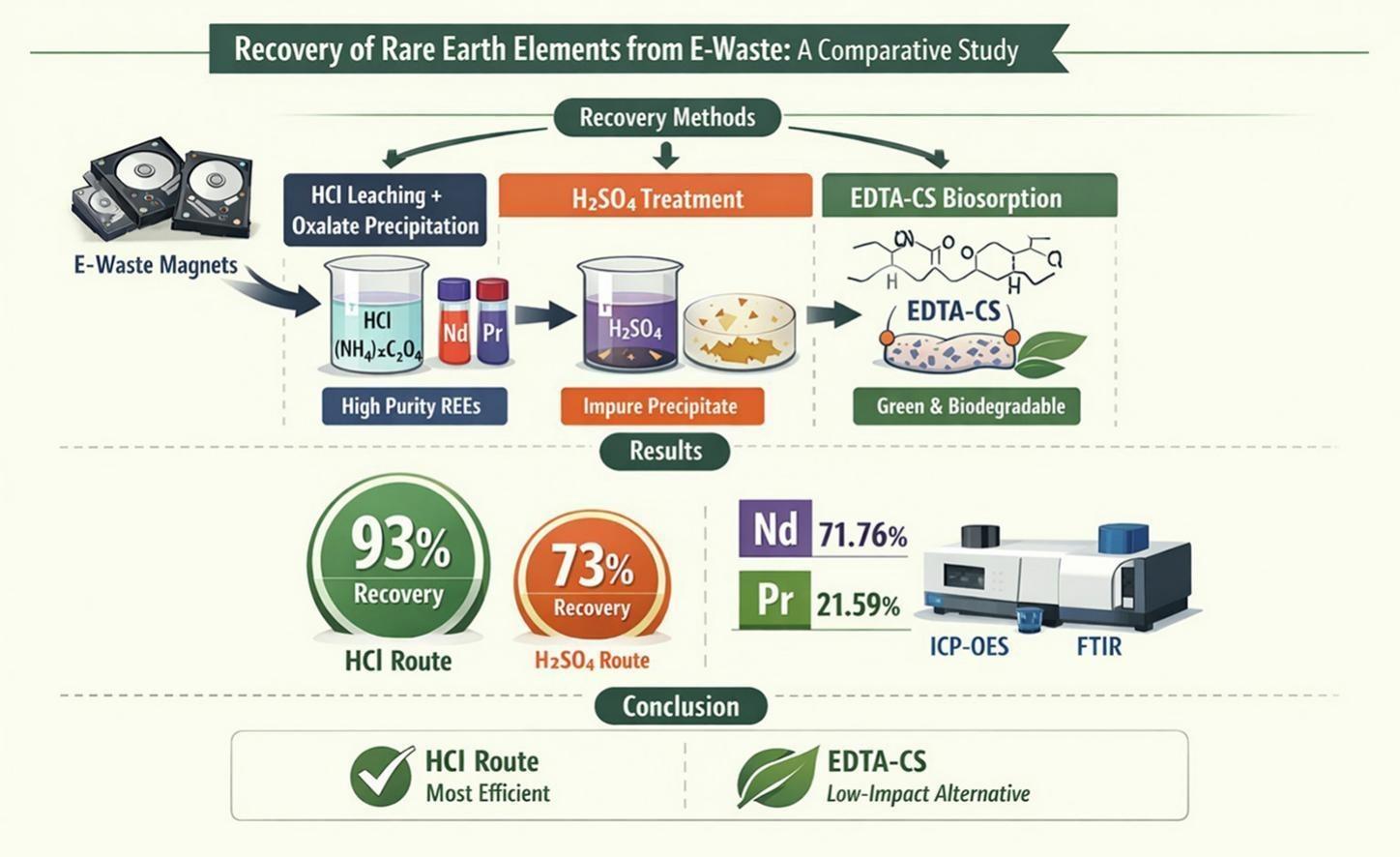
Study Design: Three Paths to Recovery
The team compared three recycling approaches under identical conditions:
(1) Hydrochloric acid (HCl) leaching followed by oxalate precipitation,
(2) Sulfuric acid (H₂SO₄) leaching, and
(3) Biosorption using EDTA-functionalized chitosan, a biodegradable polymer.
Magnets from discarded hard drives were demagnetized, stripped of coatings, crushed, and processed. Advanced tools like ICP-OES tracked recovery efficiency and impurity removal.
| Method | How it Works (technical) | Explanation | Recovery Efficiency | Pros | Cons/Challenges |
|---|---|---|---|---|---|
| Hydrochloric Acid (HCl) Leaching + Oxalate Precipitation | Magnets are dissolved in hydrochloric acid to release rare earth elements, which are then selectively precipitated out using oxalate chemistry | Dissolve the magnet in acid, then pull the valuable metals back out as a solid. | ~93% (highest) | High recovery rate; good separation from iron; well-understood chemistry | Uses strong acids; generates chemical waste; environmental handling required |
| Sulfuric Acid (H₂SO₄) Leaching | Similar to HCl method, but uses sulfuric acid to dissolve materials, followed by recovery steps | Use a different acid to break down the magnet and try to extract the metals. | ~73% (lower) | Established industrial chemical; widely available | Less efficient; more impurities due to co-precipitation; lower selectivity |
| Biosorption (EDTA-Functionalized Chitosan) | A biodegradable polymer (chitosan) modified with EDTA binds selectively to rare earth ions in solution | Use a sponge-like natural material that ‘grabs’ the valuable metals from liquid. | Not directly comparable (promising but early-stage) | Environmentally friendly; lower chemical impact; selective binding | Early-stage; scalability uncertain; slower process; less proven commercially |
Key Findings: Efficiency vs Sustainability Trade-Off
The HCl method delivered the highest recovery (93%), yielding a concentrated mix of Nd (72%) and Pr (22%) with minimal iron contamination. The sulfuric acid route lagged (73%) due to co-precipitation losses. Meanwhile, the chitosan-EDTA method offered a lower-impact, environmentally friendlier option, though less mature for industrial use.
Implications: Recycling Gains Strategic Importance
This work reinforces a growing reality: urban mining—recovering rare earths from e-waste—could become a meaningful supplement to primary supply, especially for the West seeking to reduce reliance on Chinese-controlled refining. The ability to recover high-value magnet materials efficiently is critical as demand accelerates across EVs and defense systems.
Limitations: Lab Success ≠ Industrial Reality
The study remains laboratory-scale, with limited validation using real-world mixed e-waste streams. Chemical processing at scale raises cost, environmental, and regulatory challenges, particularly around acid handling and waste management. The biodegradable route, while promising, requires further optimization.
Bottom Line
Recycling rare earths from electronic waste is no longer theoretical—it is technically viable. But turning 93% recovery in the lab into scalable, economic supply chains remains the real test.
Citation: Abbas, M.D. et al. (2026). Comparative evaluation of recovery routes for neodymium and praseodymium from discarded hard disk drive magnets. Materials Letters.

0 Comments
No replies yet
Loading new replies...
Moderator
Join the full discussion at the Rare Earth Exchanges Forum →